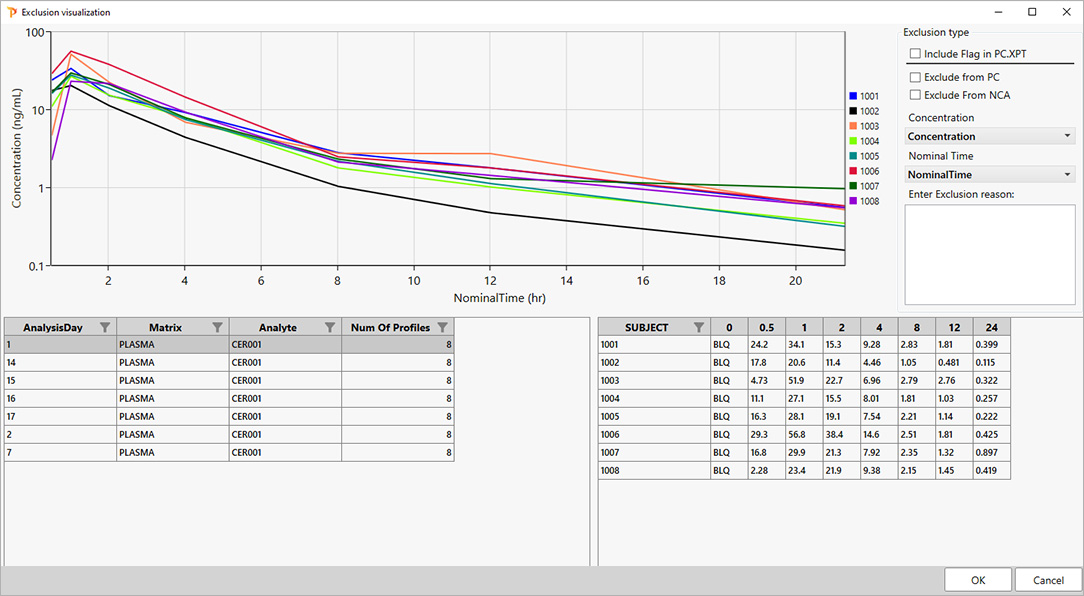
对于 PK 科学家而言,速度与准确性在药物研发生命周期的每个阶段都至关重要。Certara PK Submit® is a powerful plug-in for Phoenix®, designed to streamline non-compartmental analysis (NCA) and generate validated PK-related CDISC submission datasets.
通过自动化关键步骤并提供引导式直观界面,PK Submit™ 显著减少了 PK 数据集准备与分析的时间及复杂性。这一创新解决方案使科学家能够专注于从研究数据中提取高价值洞见,同时确保符合监管标准。