Define.xml 文件是 SDTM、SEND 及 ADaM 数据集监管提交的关键组成部分。However, creating these files manually is time-consuming and prone to errors. Pinnacle 21® Enterprise simplifies this process by automating the creation of define.xml files, dataset metadata, annotated CRFs, and Reviewer’s Guides. With built-in validation and intuitive tools, you can ensure your submissions are accurate, compliant, and ready for approval.
Pinnacle 21®
Define-XML: Automate your Define.xml files with Pinnacle 21

Learn more about automating Define-XML

Effortlessly create define.xml files for SDTM, SEND, and ADaM datasets
Pinnacle 21 Enterprise’s define-xml tool is designed to streamline your submission process, ensuring compliance and efficiency. By embedding the Define-XML standard into dataset design, the platform reduces manual effort and eliminates costly errors.
Automate define.xml
Automatically create define.xml files for SDTM, SEND, and ADaM. Simply edit and manage files like a spreadsheet! You can also import spreadsheets to instantly convert to XML format.
Accurately predict define.xml
When you design forms for an EDC in P21E, the platform accurately predicts the define.xml for the datasets that would be created – based on your form design. That way you’ll have submission deliverables from study startup.
Define.xml 可视化
Preview your define.xml metadata in PDF, or using the published CDISC stylesheet. This includes full definition of the define, page links to annotated CRFs, and hyperlinks to submission datasets.
实时验证确保合规
任何错误或无效条目都会立即实时高亮显示。 So validation is baked in from the start, and you can be confident your define.xml is flawless! Even import existing spreadsheets, SDTM or ADaM datasets into the platform and validate in real-time. All that’s left is to extract your final define.xml file at the click of a button!
Manage version control
You can update and version your Define.xml files in P21E, allowing complete visibility and change control. Assess changes over time by comparing studies, study versions, or comparing content with standards.
Generate Reviewer’s Guides
Submit clear, complete data and easily share context and pre-empt questions in P21E. Our tool automates content aggregation and formatting, allowing further time savings. Then it’s just one click to generate your submission ready Reviewer’s Guide!
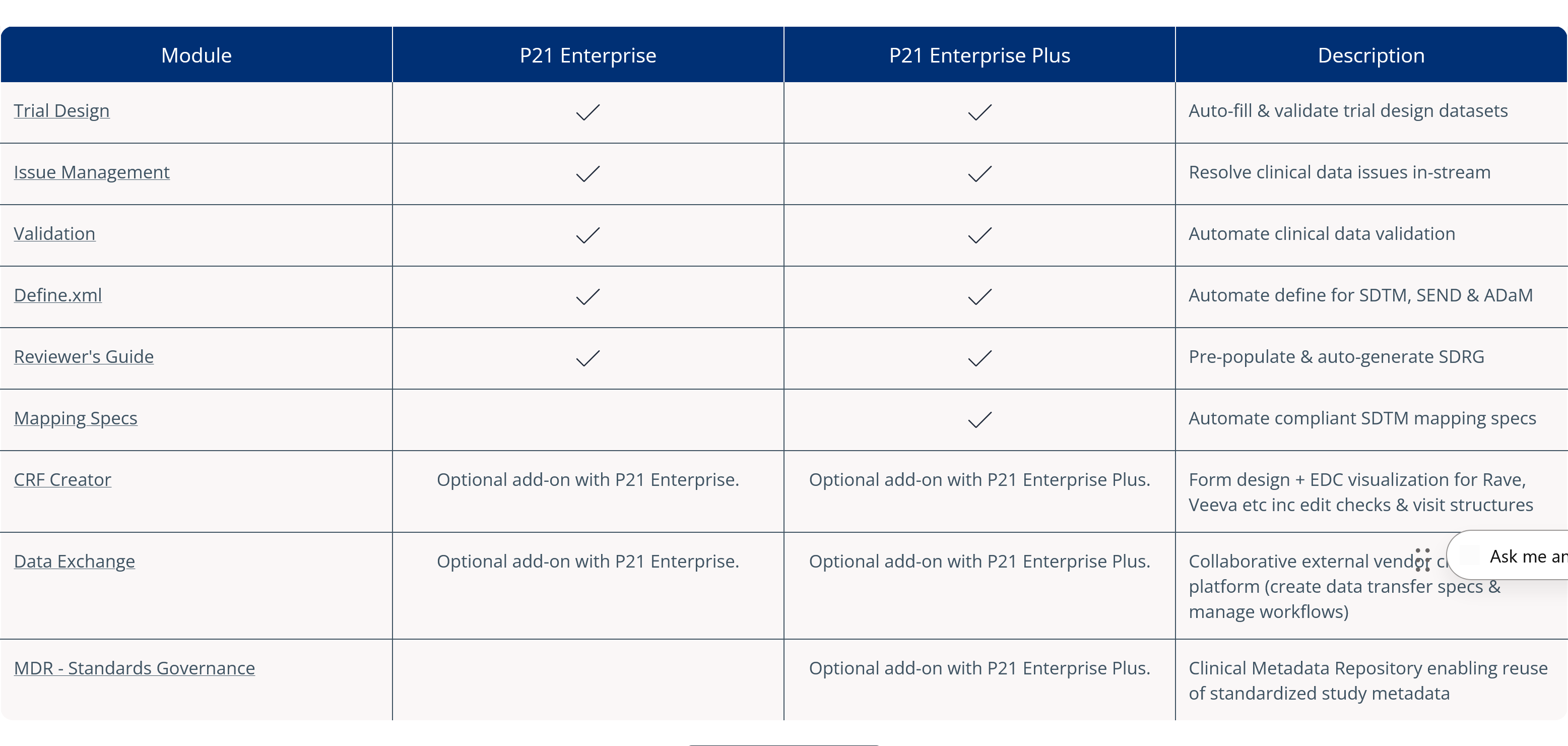
Pinnacle 21 Enterprise Software Suite
With Pinnacle 21 Enterprise as your foundation, add additional functionality according to your requirements. Get in touch to find out more about adding additional modules to your Enterprise platform.
如何更好地使用 Pinnacle 21 Enterprise
相关产品
Empowering CROs to improve sponsor service and outcomes with Pinnacle 21 Enterprise
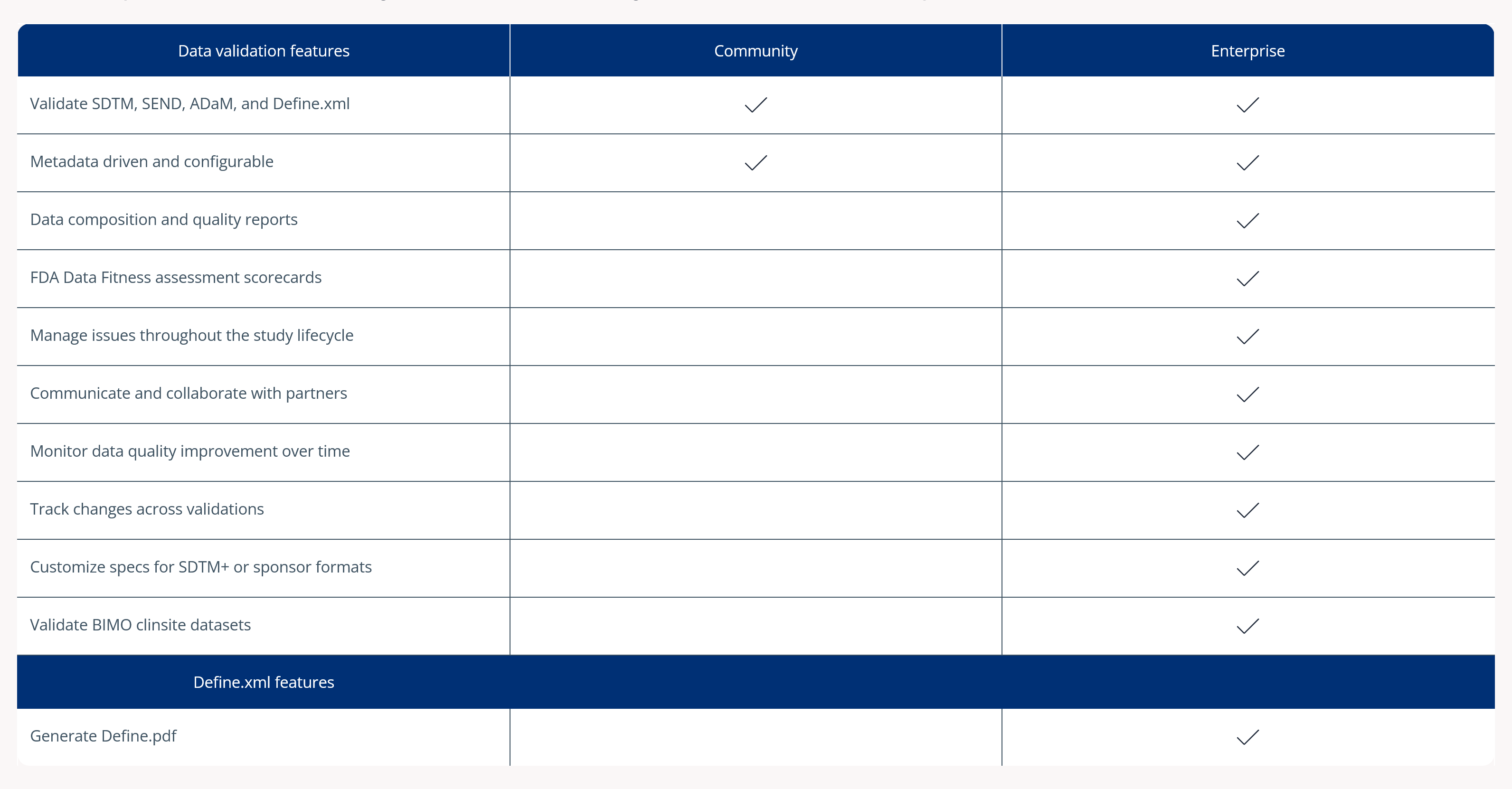
Delivering accurate Define.xml packages goes beyond the limitations of Community. Sponsor submissions require early error detection, continuous validation, and version control to avoid costly end-of-study rework and delays.
Pinnacle 21 Enterprise provides a centralized, submission-ready framework that allows CROs to automate flawless submission deliverables at scale.
Technical support with define.xml
Got an urgent deadline or need a hand producing your define.xml submission package? Our experienced technical experts are here to help prepare your submission ready data, ensuring quality and compliance.
为什么选择 Certara?
Certara empowers drug development companies and professionals with innovative solutions that help ensure compliance, accelerate timelines, and enhance outcomes.
Trusted by the FDA and PMDA, Pinnacle 21 Enterprise instills confidence that your clinical data is submission-ready. With unmatched CDISC expertise and years of experience in clinical data standards, we deliver industry-leading professional services and support to customers across the globe.

相关资源
查看全部
With Pinnacle 21, your data is secure
Certara 信息安全管理体系(ISMS)已通过 ISO 27001 认证。我们已实施严密的安全控制措施,完成严格的风险评估,并持续改进。Pinnacle 21 ensures full compliance with global data protection standards, offering peace of mind for sensitive analysis.
预约免费演示
Discover how Pinnacle 21 Enterprise can transform your clinical trial submission process. Book a no-obligation demo today to explore the benefits of automated define.xml creation.
Make an inquiry about Pinnacle products
Not ready for a demo?
Fill out this form to make an inquiry about the Pinnacle 21 product portfolio, or to discuss your requirements with our team, and we’ll get back to you right away.
常见问题解答
什么是 Define.XML?
Define.xml is the metadata file that describes the content and formatting of clinical trial submission datasets. It is a required component for regulatory submissions.
什么是 Define-XML 标准?
Define-XML 标准基于 CDISC 运行数据模型 (Operational Data Model, ODM) XML 模式。It ensures vendor-neutral and platform-independent data exchange.
Why is define.xml important?
Define.xml ensures that regulatory agencies can understand and review your clinical trial data accurately and efficiently.
Where can I download Pinnacle 21 community?
If you’re new to Pinnacle 21 and want to get a feel for its basic validation capabilities, you can access Community here: https://help.pinnacle21.certara.net/en/articles/9736610-download-pinnacle-21-community