2026 年 4 月 9 日
Why PK parameters matter
PK parameters are not “just statistics.”They are a compact language for exposure, often the most measurable bridge between what you administer, such as dose, formulation, or regimen, and what the body experiences in terms of systemic drug concentration over time. Regulators and development teams use pharmacokinetic (PK) metrics to compare products in bioequivalence (BE) assessments, predict and manage drug–drug interactions (DDIs), justify dosing in special populations, and interpret whether a formulation or dosing regimen change is likely to have clinical relevance.
The concentration time curve as the foundation of PK parameters
At the center is the concentration–time curve. From this curve we extract exposure and timing descriptors. The EMA explicitly frames BE around the plasma concentration–time curve, where AUC reflects the extent of exposure, while Cmax and tmax are influenced by absorption rate.
The essential PK parameters and how to read them correctly
| wdt_ID | wdt_created_by | wdt_created_at | wdt_last_edited_by | wdt_last_edited_at | Parameter | Definition (plain language) | Mathematical intuition (very short) | Biological meaning (typical) | Development relevance | Common misconceptions |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | DaniellePillsbury | 09/04/2026 05:02 PM | DaniellePillsbury | 09/04/2026 05:02 PM | Cmax | Maximum observed concentration | Sample-based maximum; sensitive to early sampling | Proxy for peak systemic burden | BE endpoint; peak-driven safety concerns in some settings | “Higher Cmax always means better efficacy” (depends on exposure–response) |
| 2 | DaniellePillsbury | 09/04/2026 05:03 PM | DaniellePillsbury | 09/04/2026 05:03 PM | Tmax | Time to reach Cmax | Argmax time; unstable with sparse sampling | Absorption rate influenced timing | Often supportive (not always required statistically) | “Different Tmax always means non-equivalence” |
| 3 | DaniellePillsbury | 09/04/2026 05:04 PM | DaniellePillsbury | 09/04/2026 05:04 PM | AUC | Area under concentration–time curve | Integral (often trapezoids in NCA) | Overall systemic exposure | BE endpoint; basis for many dose/exposure discussions | “AUC tells you mechanism” (it’s integrative, not mechanistic) |
| 4 | DaniellePillsbury | 09/04/2026 05:05 PM | DaniellePillsbury | 09/04/2026 05:05 PM | Half-life (t½) | Time for concentration to halve in terminal phase | t½ = 0.693·V/CL (linear) | Joint signal of distribution + elimination | Washout, accumulation, dosing interval planning | “Long t½ means low clearance” (could also be high V) |
| 5 | DaniellePillsbury | 09/04/2026 05:06 PM | DaniellePillsbury | 09/04/2026 05:06 PM | Clearance (CL) | “Volume” cleared per time | CL = Rate_elim / C; also CL = F·Dose/AUC | Elimination capacity (integrating organs/processes) | Core for dose scaling and DDI/subpopulation impact | “CL is how much drug is removed per hour” (it’s a proportionality) |
| 6 | DaniellePillsbury | 09/04/2026 05:07 PM | DaniellePillsbury | 09/04/2026 05:11 PM | Volume of distribution (Vd) | Apparent volume relating amount to concentration | Vd ≈ Amount / C (conceptually) | Extent of distribution outside plasma | Loading dose logic; interpreting t½ shifts | “Vd is an anatomical volume” (it’s an apparent/theoretical one) |
Cmax: The peak plasma level a drug reaches
- Definition: Cmax (maximum observed concentration) is the highest measured drug concentration in the sampled concentration time profile, often in plasma or serum.
- Mathematical intuition: Cmax is a function of both the true peak and the sampling schedule. FDA explicitly notes that if the first sample is the highest point, Cmax may be biased due to insufficient early sampling.
- Biological meaning: Often a proxy for peak systemic burden; sometimes relevant for acute adverse events or rapid-onset pharmacology (context dependent).
- Development relevance: In EU BE, Cmax is a required parameter (with AUC) for single-dose studies.
- Common misconceptions: “Cmax is the efficacy metric.”E–R guidance stresses that clinical decisions need exposure–response understanding, not a single PK number.
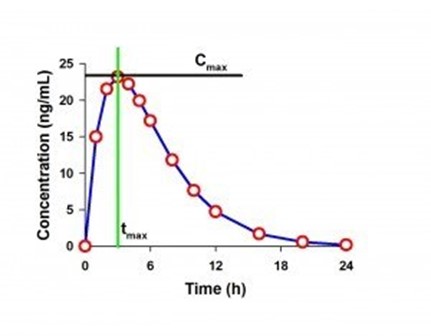
Fig.: Example PK Curve with Cmax and tmax labeled
Tmax: Capturing the speed of absorption
- Definition: Time to reach Cmax.
- Mathematical intuition: Tmax is discrete (depends on sampling times), making it noisy and sometimes non-symmetric statistically.
- Biological meaning: Influenced by absorption rate; in BE contexts, EMA notes Cmax and tmax are influenced by absorption rate.
- Development relevance: EMA states statistical evaluation of tmax is not required, but if rapid release is clinically important (onset or adverse events), there should be no “apparent” difference in median tmax and its variability.
- Common misconceptions: “Different Tmax always fails BE.”In many cases, it is supportive rather than primary.
AUC: Measuring total drug exposure over time
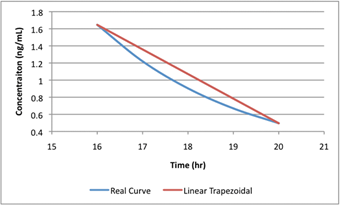
Fig.: Linear AUC assumes straight lines; real curves are not.
- Definition: Area under the concentration–time curve, i.e., total exposure over time.
- Mathematical intuition: AUC is an integral; in practice NCA uses trapezoidal approximations between observed points, plus a terminal extrapolation if estimating AUC∞.
- Biological meaning: In linear kinetics, AUC increases with dose and decreases with clearance.
- Development relevance: EMA specifies AUC(0–t) (or, when relevant, AUC(0–72h)) and Cmax for single-dose BE, with 90% CI acceptance 80.00–125.00%.
- Common misconceptions: “AUC captures absorption speed.”AUC is primarily an extent-of-exposure metric; rate can change Cmax/Tmax without changing AUC.
Half-life (t½): Understanding drug persistence over time
- Definition: Time for concentration to fall by half during the terminal phase (under linear/first-order assumptions).
- Mathematical intuition: t½ = 0.693/ke and, because CL = ke·Vd, also t½ = 0.693·Vd/CL.
- Biological meaning: Not a “pure elimination” metric – distribution (Vd) and elimination (CL) both matter.
- Development relevance: Guides washout length, sampling duration, and accumulation expectations for repeated dosing (regimen design).
- Common misconceptions: “Long half-life implies organ failure or low CL.”Half-life can increase if Vd increases even at constant CL.
Clearance (CL): The body’s capacity to eliminate drug
- Definition: “Rate of drug elimination divided by plasma concentration,” yielding an apparent plasma volume cleared per unit time.
- Mathematical intuition: In linear kinetics, CL links dose and exposure: AUC = F·Dose/CL (so CL = F·Dose/AUC).
- Biological meaning: Summarizes elimination rate across pathways (renal/hepatic/other).
- Development relevance: A primary driver of dose requirements and a key object/perpetrator concept in DDI workstreams (changes in CL often explain AUC changes).
- Common misconceptions: “CL tells you how much drug is removed per hour.”It is a proportionality; mg/h depends on concentration and CL.
Volume of distribution (Vd): How far a drug travels beyond the bloodstream
- Definition: An apparent/theoretical volume indicating the extent of distribution relative to plasma concentration.
- Mathematical intuition: Conceptually, Amount ≈ C·Vd (so Vd ≈ Amount/C).
- Biological meaning: A high volume of distribution typically indicates extensive distribution into tissues (or binding), not a “bigger body.”
- Development relevance: Influences loading-dose logic and helps interpret half-life changes (since t½ depends on Vd and CL).
- Common misconceptions: “Vd is anatomical volume.”It is explicitly described as apparent/theoretical.
Looking to go beyond the basics of PK parameters?
Authored by Prof. Johan Gabrielsson, the trusted reference book Pharmacokinetic and Pharmacodynamic Data Analysis provides comprehensive insights into pharmacokinetics, pharmacodynamics, and PK/PD concepts.
Register with your professional or academic email to access the full standard edition – completely free.
Linking dose, clearance, and exposure: The core PK equations
| wdt_ID | wdt_created_by | wdt_created_at | wdt_last_edited_by | wdt_last_edited_at | Concept | Key Equation | What It Shows | Why It Matters |
|---|---|---|---|---|---|---|---|---|
| 1 | DaniellePillsbury | 09/04/2026 06:27 PM | DaniellePillsbury | 09/04/2026 06:29 PM | One compartment IV bolus model | C(t) = C0 · e^(−ke · t) C0 = Dose / Vd | Concentration declines exponentially after IV bolus dosing | Foundation for deriving exposure relationships |
| 2 | DaniellePillsbury | 09/04/2026 06:29 PM | DaniellePillsbury | 09/04/2026 06:29 PM | AUC derivation (IV) | AUC(0→∞) = ∫₀^∞ C(t) dt = C0 / ke = (Dose / Vd) / ke | Total exposure equals initial concentration divided by elimination rate constant | Connects exposure directly to elimination |
| 3 | DaniellePillsbury | 09/04/2026 06:29 PM | DaniellePillsbury | 09/04/2026 06:29 PM | Clearance relationship | CL = ke · Vd → AUC = Dose / CL (IV) | AUC is inversely proportional to clearance | Decreased clearance increases exposure |
| 4 | DaniellePillsbury | 09/04/2026 06:29 PM | DaniellePillsbury | 09/04/2026 06:29 PM | Extravascular dosing | AUC = F · Dose / CL | Exposure depends on bioavailability and clearance | Explains oral dosing and DDI effects |
| 5 | DaniellePillsbury | 09/04/2026 06:30 PM | DaniellePillsbury | 09/04/2026 06:30 PM | Half life relationship | t½ = 0.693 / ke → t½ = 0.693 · Vd / CL | Half life depends on both distribution and clearance | Long half life does not automatically mean low clearance |
| 6 | DaniellePillsbury | 09/04/2026 06:30 PM | DaniellePillsbury | 09/04/2026 06:31 PM | Steady state average concentration | Cavg,ss = (F · Dose) / (CL · τ) | Average exposure over a dosing interval depends on dose, clearance, and interval | Guides regimen design and dose adjustments |
Numeric intuition builders
| wdt_ID | wdt_created_by | wdt_created_at | wdt_last_edited_by | wdt_last_edited_at | Scenario | Inputs | Result | Interpretation |
|---|---|---|---|---|---|---|---|---|
| 1 | DaniellePillsbury | 09/04/2026 06:27 PM | DaniellePillsbury | 09/04/2026 06:35 PM | Example A: Clearance decreases | Dose = 100 mg CL = 5 L/h | AUC = 100 / 5 = 20 mg·h/L | Baseline exposure |
| 2 | DaniellePillsbury | 09/04/2026 06:29 PM | DaniellePillsbury | 10/04/2026 12:10 PM | Example A (continued) | CL decreases to 2.5 L/h | AUC = 100 / 2.5 = 40 mg·h/L | Halving clearance doubles exposure. Explains DDI or organ impairment effects |
| 3 | DaniellePillsbury | 09/04/2026 06:29 PM | DaniellePillsbury | 09/04/2026 06:36 PM | Example B: Absorption slows | Same Dose and CL Slower absorption rate | AUC similar Cmax lower Tmax later | Rate can change peak and timing without changing total exposure |
Common pitfalls in translating PK parameters into decisions
A common pitfall in pharmacokinetic interpretation is over-reliance on single PK parameters. AUC and Cmax answer different questions: AUC reflects total exposure, while Cmax reflects peak concentration. Exposure–response considerations determine which matters more in a given context, so interpreting either in isolation can mislead.
Sampling artifacts are another risk. Insufficient early sampling can produce a “first point Cmax,” biasing peak estimates, and inadequate sampling around Tmax can distort peak timing and magnitude.
Half-life is often misread as elimination speed, yet it depends on both clearance and volume of distribution (t½ = 0.693 · Vd / CL). A longer half-life does not necessarily mean reduced clearance.
AUC truncation can also be problematic. In flip-flop kinetics, where absorption drives the terminal phase, truncated AUC may misrepresent exposure.
Finally, apparent parameters should not be treated as physiological truths. The volume of distribution is a theoretical construct, not an anatomical space.
NCA vs model-based (PopPK, PBPK, MIDD): Pros/cons and when to use
PK parameters can be derived and interpreted using different analytical approaches, including NCA, PopPK, PBPK, and broader model informed drug development frameworks. Each method uses PK parameters differently, ranging from descriptive summaries to mechanistic prediction. Choosing the right approach depends on the development and regulatory question at hand.
NCA (noncompartmental analysis)
- Pros: fast, transparent, widely accepted for standard BE endpoints; EMA requires NCA and rejects compartmental methods for BE parameter estimation.
- Cons: limited extrapolation; sparse sampling and complex kinetics can make terminal slope (and thus AUC∞, half-life) fragile.
PopPK (population PK)
- Pros: quantifies variability and covariate effects, supports individualized dosing and labeling; FDA describes PopPK as frequently used to guide development and therapeutic individualization and gives expectations for submissions and report content.
- Cons: requires stronger assumptions and careful diagnostics; EMA stresses clarity of assumptions/decisions to enable secondary evaluation.
PBPK (physiologically based PK)
- Pros: mechanistic; useful for DDI prediction and dose selection (e.g., pediatric, first-in-human) and supports “what if” simulations; EMA defines PBPK and notes these common regulatory purposes.
- Cons: platform and model qualification and documentation burden is high; EMA explicitly notes platforms need qualification for intended use when supporting regulatory decisions. FDA provides expectations for submitting PBPK analyses (format/content).
MIDD (model-informed drug development)
A forward-looking umbrella: ICH M15 provides general principles and a harmonized evidence framework; the EMA Step 5 page lists a legal effective date of 23 July 2026 (after today).
How software enables robust PK parameter assessment
PK parameters may be conceptually straightforward, but their accurate calculation depends on validated analytical software. In modern drug development, platforms such as Phoenix WinNonlin® are widely used to derive key PK parameters including AUC, Cmax, Tmax, clearance, volume of distribution, and half life from concentration time data.
These tools apply standardized noncompartmental methods, ensure reproducibility, and provide the traceability required for regulatory submissions. Beyond basic calculations, software environments increasingly support modeling and simulation, embedding PK parameters within broader decision frameworks.
Reliable pharmacokinetic software therefore ensures that PK parameters are calculated consistently, interpreted correctly, and confidently used in regulatory and development decisions.
Conclusion: Integrating PK parameters for robust decisions
PK parameters are decision tools, not trophies: each metric answers a specific question, and the most robust conclusions come from patterns across PK parameters, such as AUC, Cmax, half life, and clinical context, rather than from isolated numbers. Interpreting PK parameters in combination allows teams to distinguish between changes in absorption, distribution, and clearance instead of reacting to a single shifted value.
For regulator facing decisions, align your endpoints and interpretation of PK parameters with the intended use, whether bioequivalence, label changes, drug-drug interaction management, or dosing in special populations. At the same time, remain explicit about underlying assumptions such as linearity, route of administration, bioavailability, and sampling adequacy, because the meaning of PK parameters depends directly on these foundations.
Learn more about Phoenix WinNonlin
Calculate and interpret PK parameters with confidence using Phoenix WinNonlin, industry trusted platforms that deliver validated noncompartmental analysis, modeling, and scalable cloud based workflows for modern drug development.

Author

Sebastian Kuechenmeister
Senior Marketing ManagerSebastian Kuchenmeister has been a Senior Marketing Manager at Certara since 2022. He is a creative marketing professional with extensive expertise in multiple marketing disciplines, campaign management, media planning and a passion for content creation and go-to market strategies. Mr. Kuchenmeister earned a Bachelor of Arts degree in Political Science from the Humboldt University in Berlin, Germany.
预约 Phoenix 演示
准备好见证 Phoenix 实战演示了吗?通过为您量身定制的指导演示,改变您的 PK/PD 分析。我们将向您展示 Phoenix 如何为您的药物开发之旅架起桥梁。
精选推荐







