2026 年 4 月 28 日


How systems modeling optimizes oncolytic virus therapies
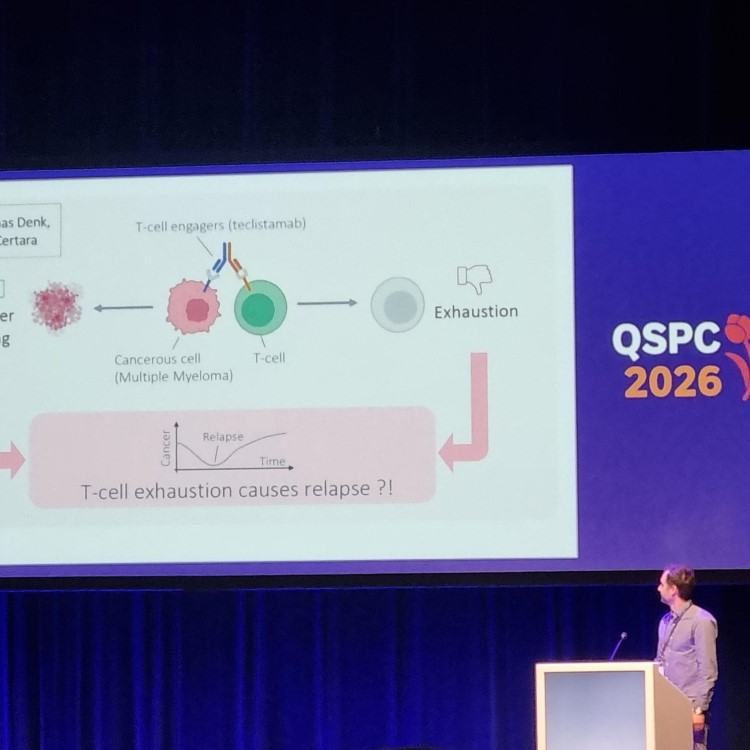
Systems modeling (QSP) helps predict how oncolytic viruses interact with tumors and the immune system, enabling smarter dosing, improved safety, and more effective cancer treatments.

Jessica Sinha
Associate Director, Marketing – QSP, CertaraJessica Sinha is an accomplished marketing leader with more than eight years of experience spanning B2B, brand, content, and digital marketing in the life sciences sector. At Certara, she leads strategic marketing initiatives for Quantitative Systems Pharmacology (QSP), combining her scientific foundation in bioengineering with a passion for clear, impactful communication.
This summary was built with significant contributions from Certara’s QSP team in attendance at QSPC 2026.
Learn more about Certara IQ
Certara IQ is the AI-enabled QSP modeling tool that will transform your research and scale your molecule’s potential.
Certara IQ offers flexible and scalable licensing options to cater to a variety of users and organization sizes.

常见问题解答
What is QSPC?
QSPC (Quantitative Systems Pharmacology Conference) is a global scientific meeting focused on advancing quantitative systems pharmacology (QSP), bringing together experts from pharma, biotech, academia, and regulatory agencies. QSPC is held every four years, making it a key milestone event for tracking long-term progress and trends in the field.
Who attends QSPC?
Attendees include:
- QSP scientists and pharmacometricians
- Drug development leaders in pharma and biotech
- Academic researchers
- Regulatory experts
What topics are covered at QSPC?
The conference typically spans:
- QSP and PBPK modeling
- 模型引导的药物开发(MIDD)
- AI/ML in MIDD approaches
Why is QSPC important for drug development?
QSPC highlights how modeling approaches are evolving from research tools into decision-making frameworks, with growing impact on regulatory submissions and clinical strategy.
See Certara IQ in Action