Preparing pharmacometrics for regulatory submissions is high-risk and time-sensitive, small gaps can lead to delays, regulatory questions, or rework.
Interactive Essential Pharmacometrics (PMx) Submission Readiness Checklist
The Certara Interactive PMx Submission Readiness Checklist
常见问题解答
What is the Interactive Essential Pharmacometrics (PMx) Submission Readiness Checklist?
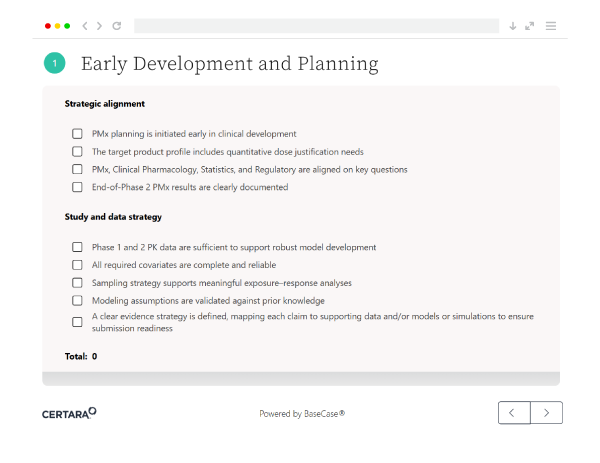
The Interactive Essential Pharmacometrics (PMx) Submission Readiness Checklistis is an interactive checklist app that helps you assess and improve your regulatory submission process. It evaluates readiness for NDA, BLA, and MAA submissions across data, modeling, documentation, and cross-functional alignment.
How is this different from a traditional checklist?
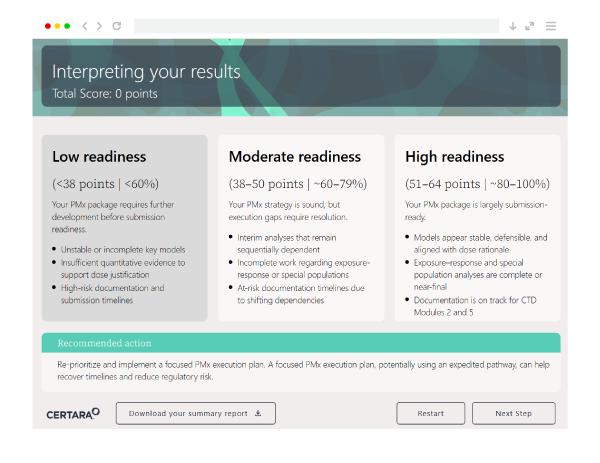
Unlike static guides, this is an interactive scorecard that provides real-time insights and scoring. It helps you identify gaps, prioritize next steps, and take action, rather than just review a checklist.
What types of submissions does this app support?
The app supports NDA, BLA, and MAA submissions, including EU MAA and rolling BLA submissions, helping teams prepare for global regulatory requirements.
Does this align with FDA and global regulatory expectations?
可以。The app is aligned with global standards, including the FDA CTD format, as well as expectations from EMA and other regulatory agencies.
What insights will I get after completing the checklist?
You’ll receive a submission readiness score, visibility into gaps across your process, and clear guidance on next steps to strengthen your submission and reduce risk.