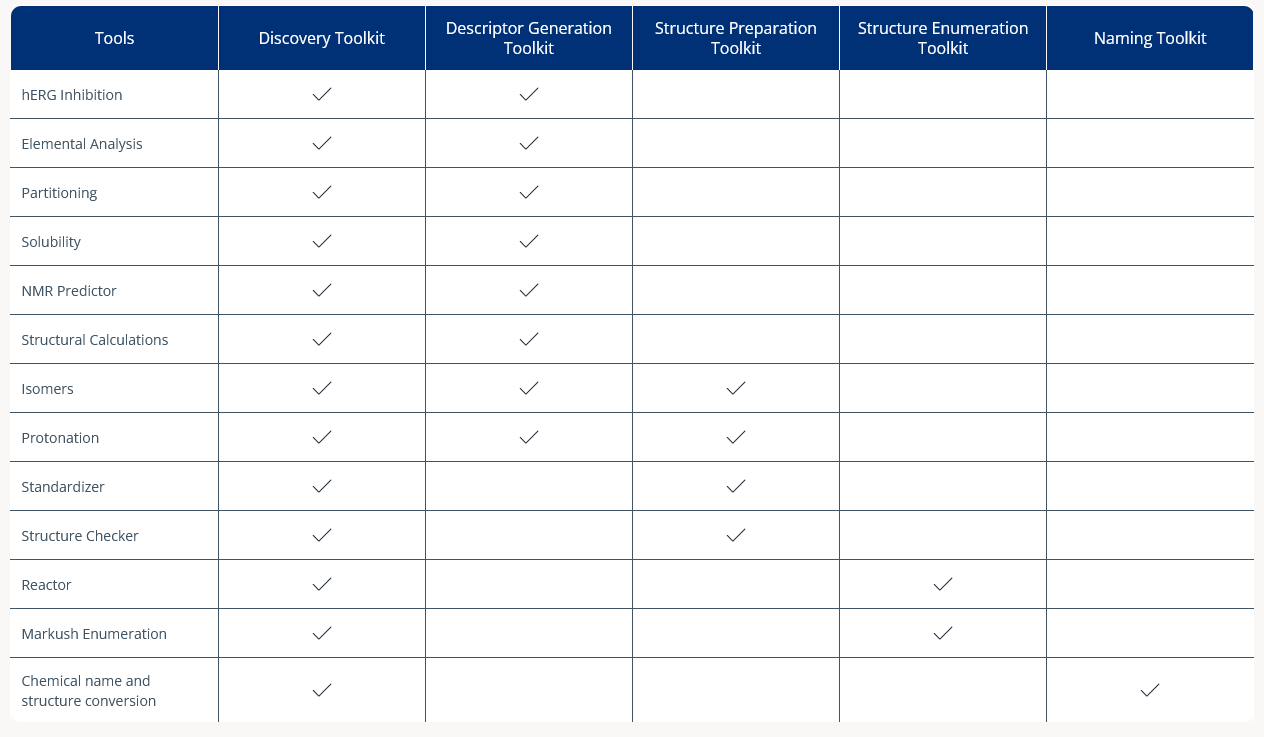
Chemical databases can get messy, making it difficult to use the data you have already gathered in chemistry research.
The Structure Preparation Toolkit allows you to validate, curate, and normalize molecules according to your business rules. This helps you prepare compounds for registration or docking, allowing you to build a unified database, and generate the relevant microspecies or isomers for your experiments more accurately based on high-quality data.