This case study features Applied BioMath’s* collaboration with Alexion Pharmaceuticals. Alexion Pharmaceuticals, Inc. is a biopharmaceutical company, focused on developing therapies for rare diseases.
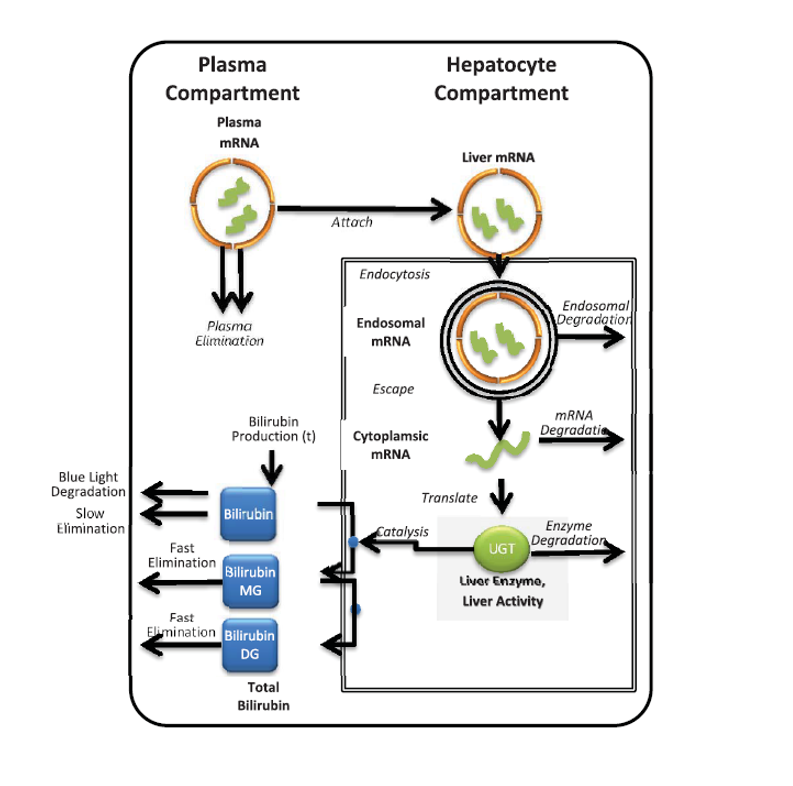
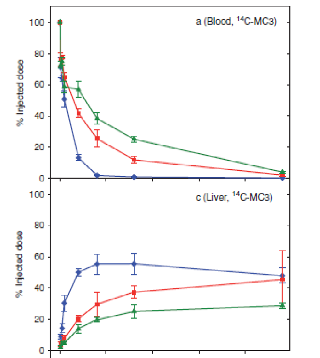
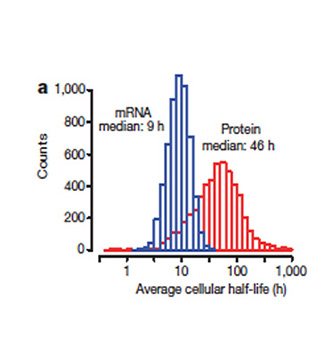
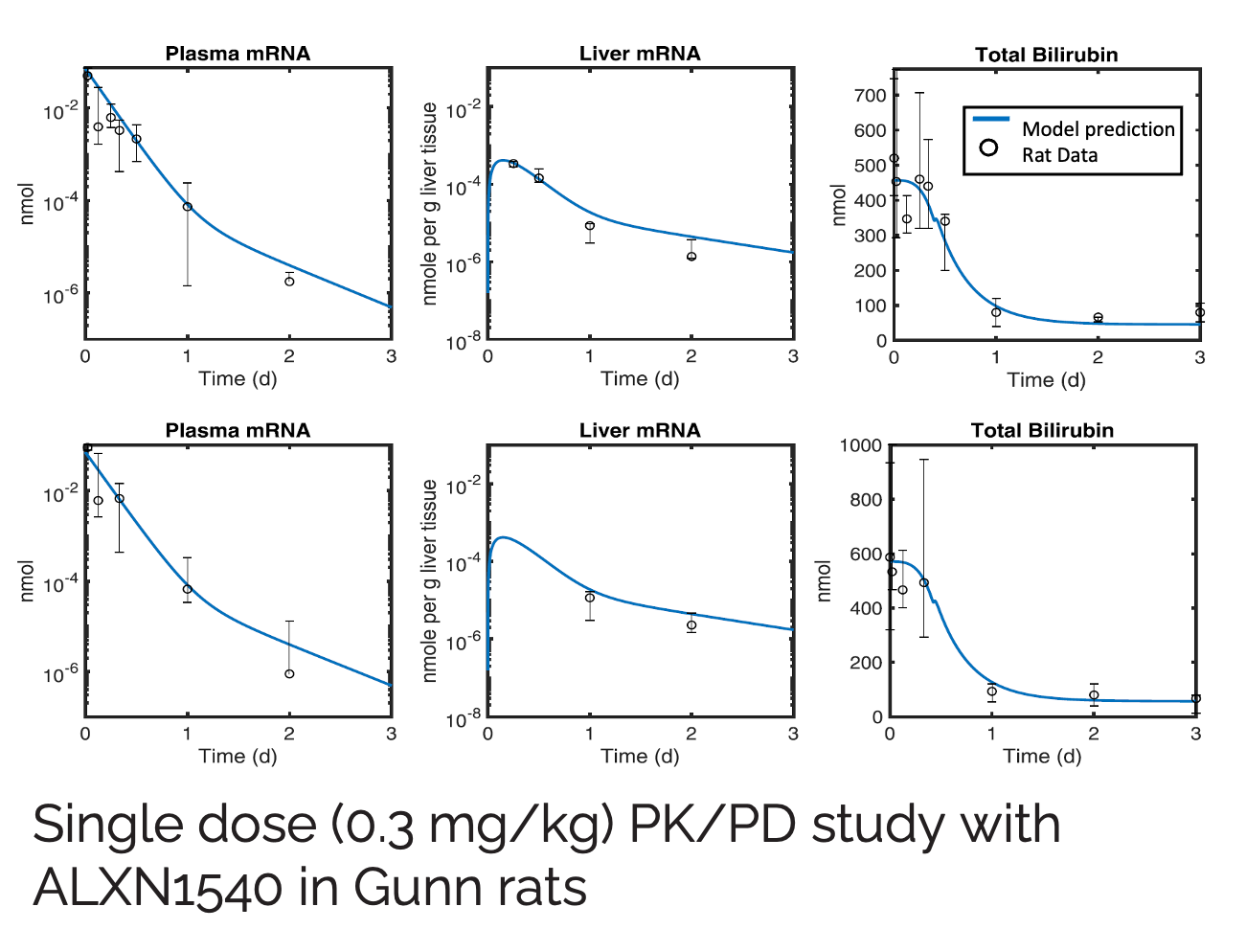
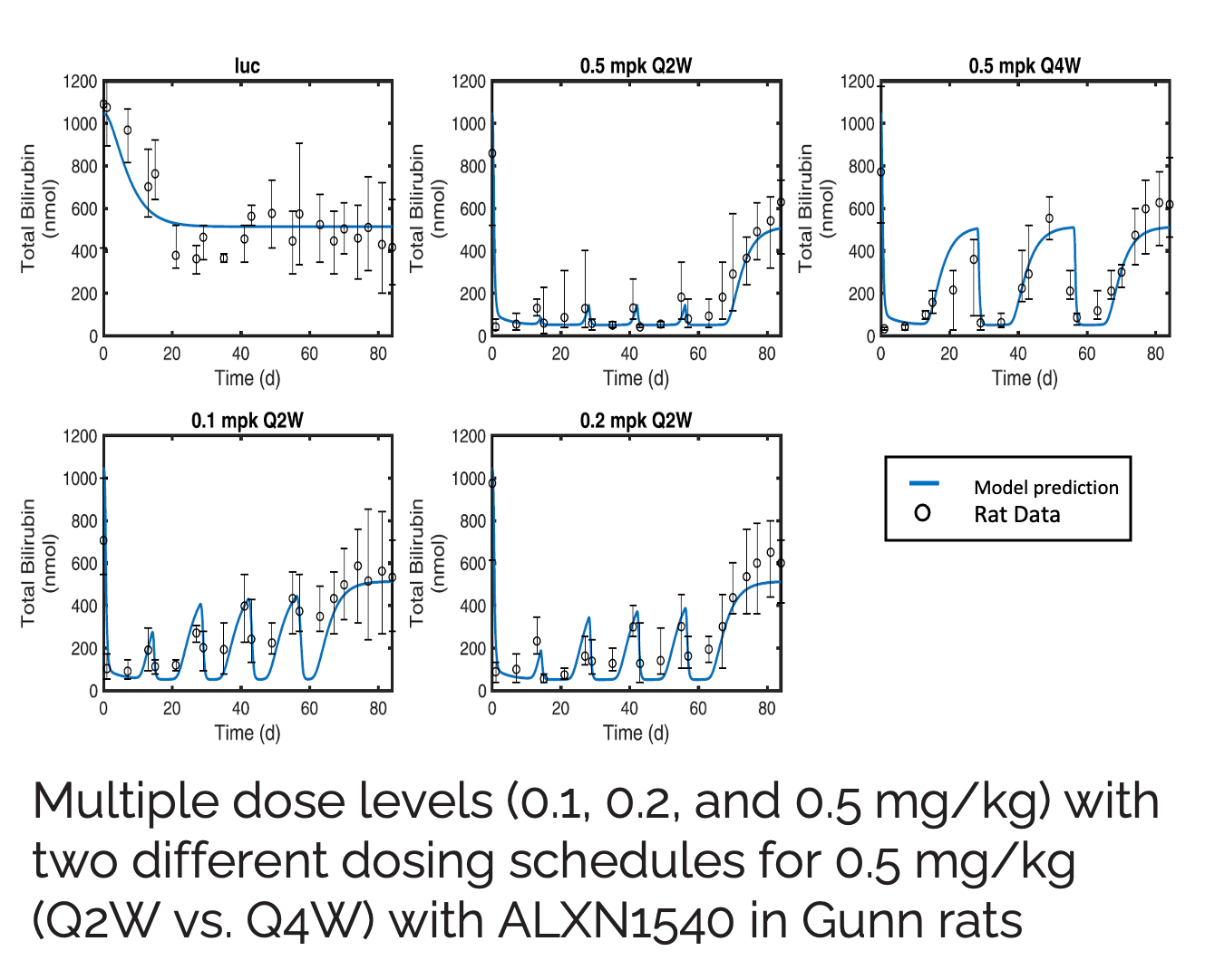
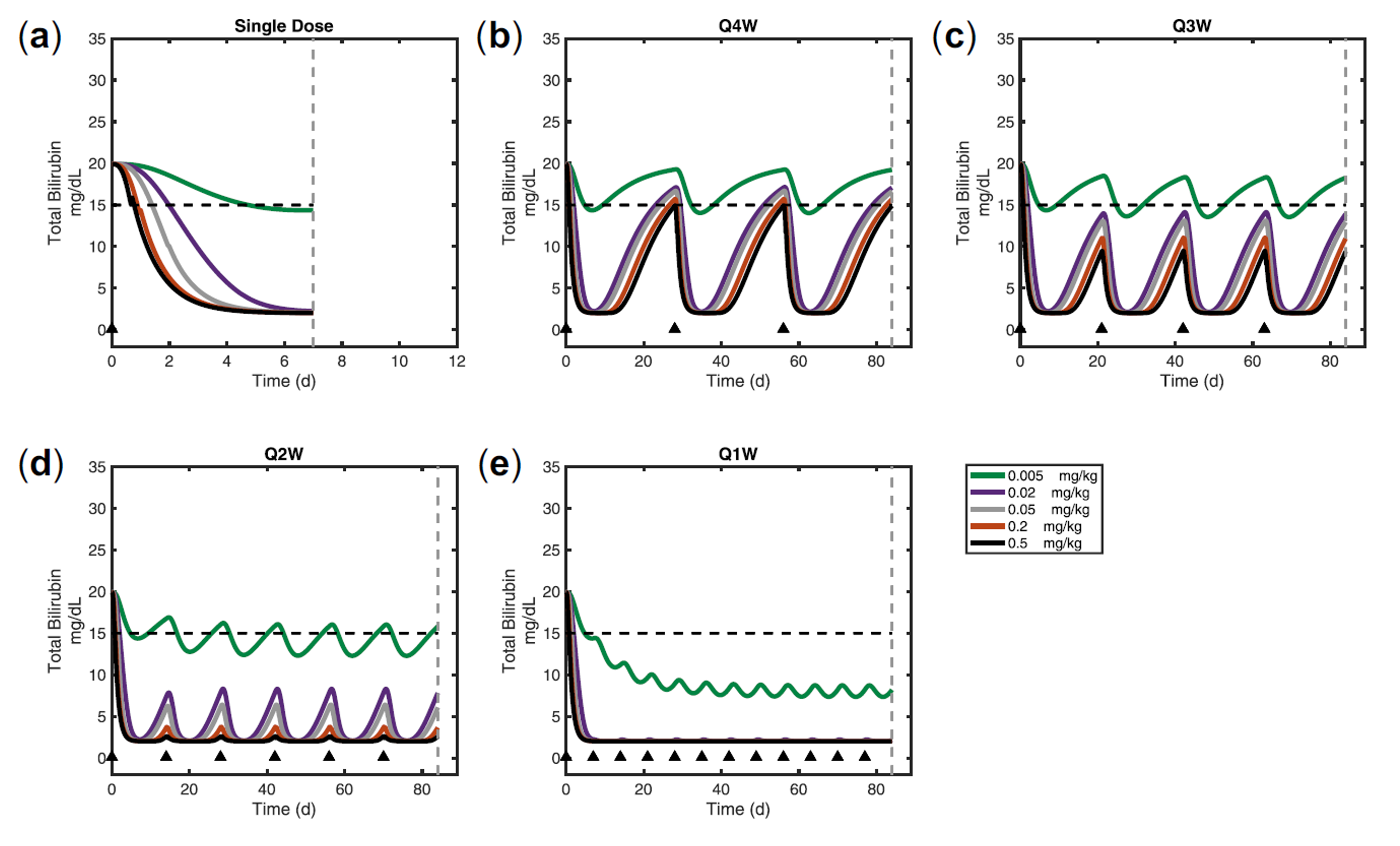
Crigler-Najjar syndrome type 1 (CN1) is an an autosomal recessive disease caused by a marked decrease in uridine-diphosphateglucuronosyltransferase (UGT1A1) enzyme activity. Delivery of a modified messenger RNA encoding for UGT1A1 (hUGT1A1-modRNA) as a lipid nanoparticle (LNP) is anticipated to restore hepatic expression of UGT1A1, allowing normal glucuronidation and clearance of bilirubin in CN1 patients. In vivo mechanism of action and clinical pharmacology is unknown.
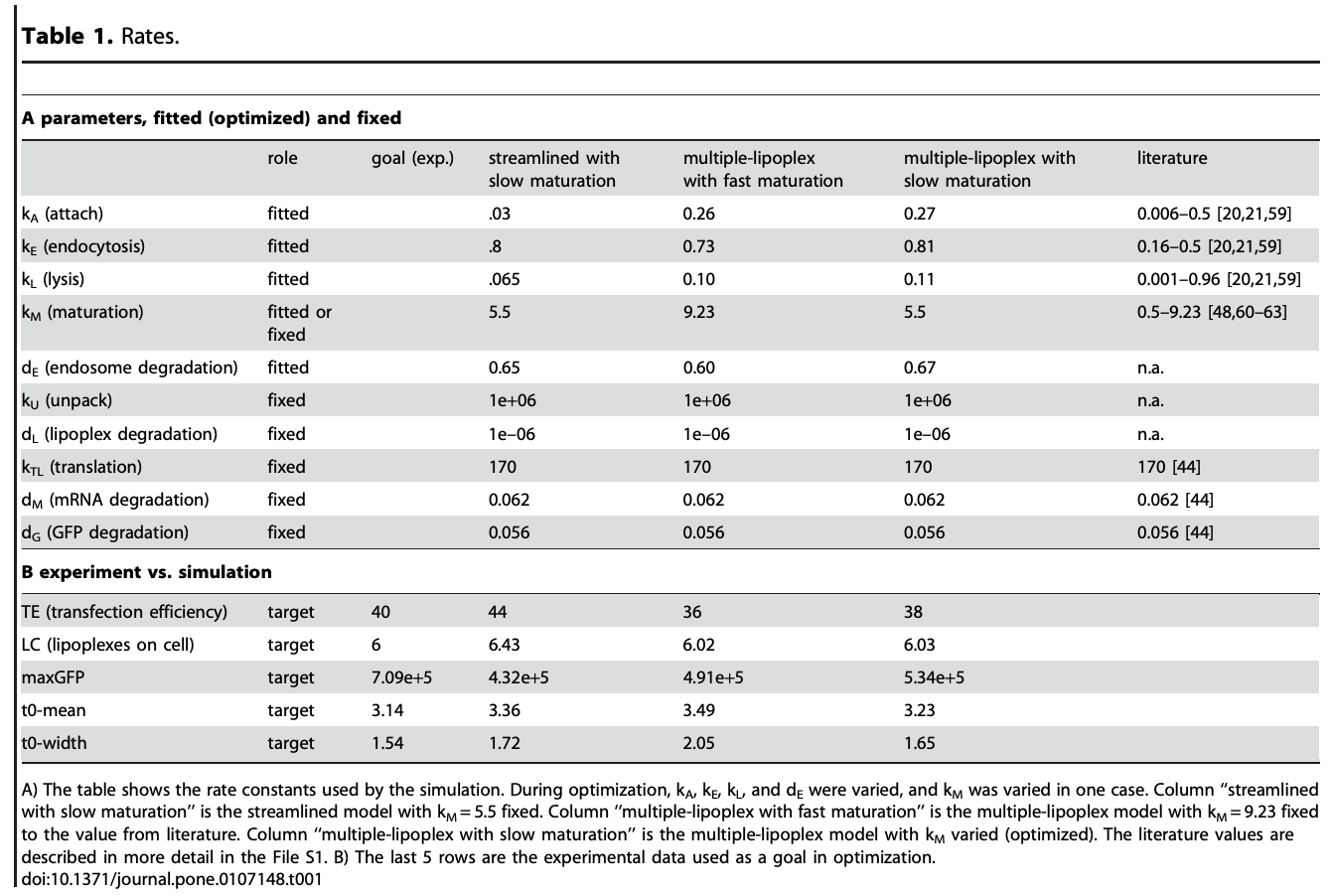
The goal of this collaboration was to develop a QSP model to support translation from preclinical to clinical studies, and first-in-human (FIH) studies and to use this QSP model to provide a deeper understanding of the mechanisms of hUGT1A1-modRNA and to guide design of the first-in-human clinical studies.
* Applied BioMath was acquired by Certara in Dec 2023.
与 QSP 专家建立联系
联系 Certara,探讨 QSP 服务如何加速您的药物研发项目我们的专家团队随时准备为您提供定制化解决方案。