2026 年 4 月 23 日
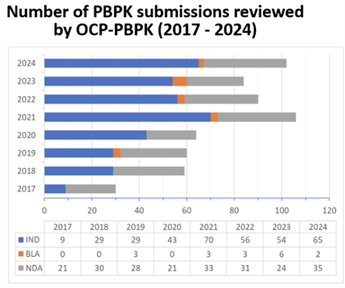
Figure 1. Number of PBPK submissions reviewed by OCP-PBPK (2017-2024) Source: Yuching, et al., DDI Marbach Workshop 2025
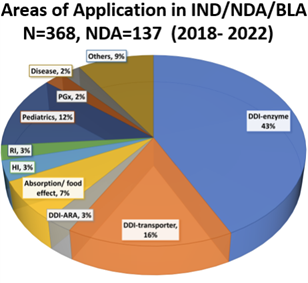
图 2. PBPK Areas of Application in IND/NDA/BLA submissions reviewed by the FDA OCP (2018-2022) Source: Yuching, et al., DDI Marbach Workshop 2025

Leverage the full potential of PBPK models to accelerate drug development
Learn how Novartis used Simcyp Simulator to support 14 FDA-approved novel drugs.
This blog was originally published in June 2021 and has been updated for accuracy.

Xian Pan, PhD
Principal Scientist, CertaraDr. Xian Pan is a Principal Scientist at Certara Predictive Technologies. She received her BSc in Pharmacy and an MSc in Pharmacokinetics from China Pharmaceutical University and earned a Ph.D. in Biopharmaceutical Sciences from the University of Illinois at Chicago. Her doctoral work focused on understanding the altered pharmacokinetics during pregnancy, specifically investigating the molecular mechanisms of CYP2D6 induction and interindividual variabilities. She has worked in the industry as a Senior Research Scientist, leading and managing preclinical DMPK projects from drug discovery to IND filing. Since joined Certara Predictive Technologies in 2017, she has significantly contributed to PBPK framework enhancement, including various modules’ development and expansion for small molecule drugs and therapeutic proteins, and evaluation of drug-drug interaction liabilities for both modalities. She has actively contributed to the expansion of the virtual populations for pediatrics, premature neonates, and fetus. She has several publications in the field and provided training and consultancy for a number of pharmaceutical companies.
Supporting PBPK applications across drug development
Simcyp offers a comprehensive suite of PBPK modeling solutions. Learn how Simcyp can support your MIDD strategies.

值得信赖的 PBPK 建模平台
预约演示或咨询,了解 Simcyp 如何为您的团队赋能
全球监管认可: 获得 11 个全球监管机构的授权许可





